Characterization of a Potential Novel Redox Partner in S. Cerevisiae Mitochondrial Disulfide Relay System and in silico Elucidating Its Essentiality
Class of 2022
Biography

- Education
- Friendship Christian School, Suwanee, GA, 2018
- The College of Wooster, BA in Biochemistry and Molecular Biology, 2022
- Professional experience
- International Student Scholarship
- MD Anderson Cancer Center, CPRIT-CURE Research Intern
- The College of Wooster, StemZone Intern, Research Assistant, Minorities in STEM secretary
IS Thesis Abstract
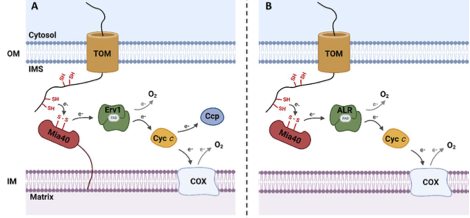
The mitochondrial disulfide relay system is a conserved pathway in eukaryotes. Via two essential components Mia40 and Erv1, the system drives the import of cysteine-rich proteins from the cytosol to the mitochondrial intermembrane space and aids their oxidative folding. Since cytochrome c, the downstream electron receptor of the pathway, is non-essential in S. cerevisiae, this suggests unknown redox components and a level of redundancy in the system. In most eukaryotic organisms, the pathway is essential thanks to its substrates’ involvement in the respiratory chain. However, this is not sufficient in explaining the pathway’s essentiality in Saccharomyces cerevisiae, as this single-celled eukaryote can survive and grow under anaerobic conditions, suggesting novel metabolic functions. Via cross-linking and co-immunoprecipitation experiments, our lab previously identified the flavodoxin-like protein Ycp4 cross-links with Erv1 in vivo. Thus, we sought to characterize Ycp4 in the context of the disulfide relay system. Here, we demonstrate that Ycp4 forms disulfide-based homodimers and re-oxidizes reduced Mia40 in vitro. Our findings suggest that Ycp4 is a non-essential redundant component of the pathway. To study the mechanism of its essentiality, we engaged an in silico approach in which we used flux balance analysis on two well-curated yeast genome-scale metabolic networks in a series of in silico knock-out experiments. We found that the essentiality of the mitochondrial disulfide relay system is connected to two other mitochondrial protein transport machineries: TIM22 and TIM23 pathways. Altogether, both approaches reveal different insights into the intricacy of this protein transport system.